
Research
Fundamental Research
The Electrochemical Research Group investigates the fundamental mechanisms and theoretical foundations of electrochemical reactions, focusing on electrochemical interfaces, catalysis, and proton-coupled electron transfer. Through advanced theoretical modelling and computational methods, the group explores the influence of interface structures on reaction mechanisms, providing insights for material design and optimization in energy and environmental applications.
Research Field
- Interfacial Electric Double Layer
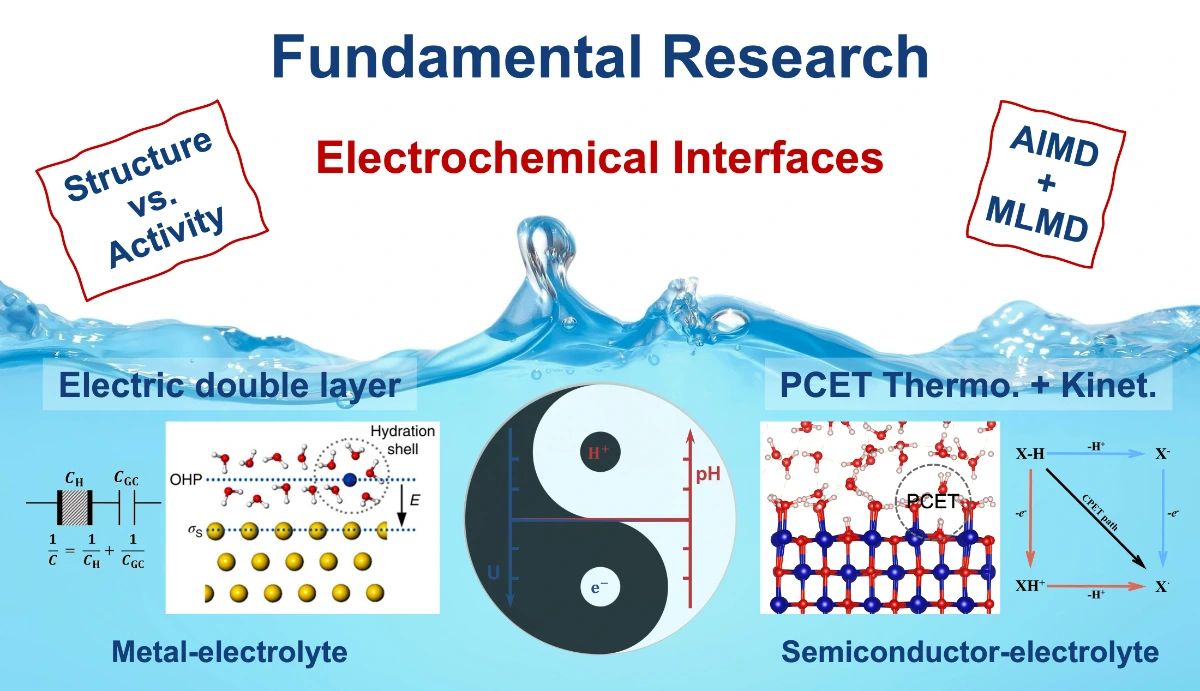
Theoretical insights into the electric double layer at solid-liquid interfaces reveal its structure, formation mechanisms, charge distribution, as well as their impact on electrochemical kinetics and catalysis. - Electrochemical Interfacial Catalytic Mechanisms
Theoretical insights into electrocatalytic mechanisms at electrochemical interfaces reveal how interfacial structures and catalyst properties dictate reaction activity, selectivity, and pathways, identifying critical steps and kinetics. - Theoretical Developments in Electrochemistry
The development of electrochemical theory establishes precise computational models for the quantitative prediction of reaction mechanisms and interfacial behaviour. - Proton-Coupled Electron Transfer(PCET)
The thermodynamic (redox potential, pKa) and kinetic (k) properties of PCET are investigated to elucidate the mechanisms of electron and proton transfer and to identify the thermodynamic constraints and kinetic barriers in catalytic systems.
Computational Electrochemistry Methods
- Computational Standard Hydrogen Electrode (cSHE)
- Redox Potential
- Acidity Constant (pKa)
- Proton-Coupled Electron Transfer (PCET) Rate Constant
- Finite Field Method
Future Perspectives
- AI-Driven Research on Electrochemical Reaction Mechanisms
- Interdisciplinary Integration and Methodological Innovation
- International Academic Collaboration and Scholarly Exchange
- AI-Driven Application Technology Transformation
Members:

程俊 
胡晋媛 
王飞腾 
朱嘉欣 
顾健 
韩思源