
Dynamic Catalysis
Heterogeneous catalysis systems exhibit highly dynamic behaviours under reaction conditions. The dynamic evolution of catalyst structures poses a great impact on catalytic performances. Traditional theoretical studies mostly focus on the most stable or several metastable catalyst structures. Further understanding of the in-situ dynamic evolution of catalysts is of great significance for understanding the reaction mechanism and optimizing the reaction activity. In previous works, our group systemetically investigated the interaction between the structural dynamics of catalysts and elementary reactions using the AIMD and free energy calculation methods. Furtherly, we developed ai2-kit workflow to generate machine learning potentials with the accuracy of first-principles calculations. This facilitates further investigation into the surface premelting of nanosized clusters and the impacts of confined supports during catalytic reactions. Looking ahead, our group will employ effective AI methods to simulate more realistic complex catalytic systems, with the aim of providing deeper insights into in-situ dynamic catalytic processes at atomic scale and facilitating the rational design and screening of catalysts by high-throughput calculations.
Research Areas
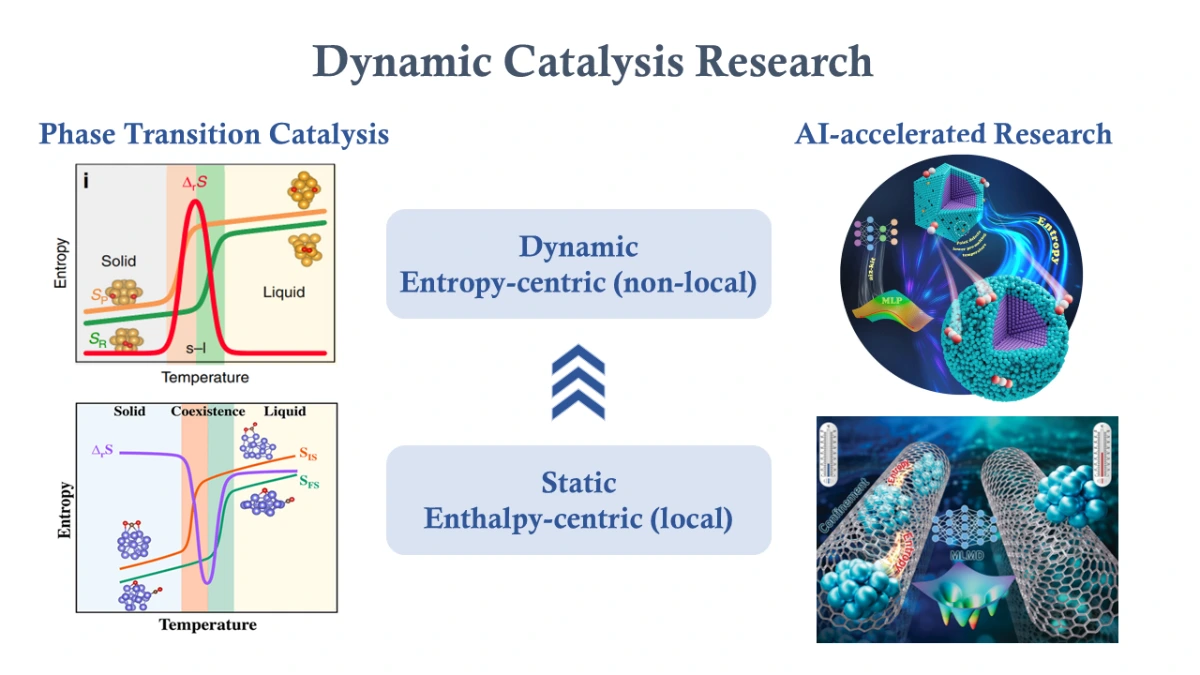
- Phase Transition Catalysis
The morphology of catalysts undergoes continuous evolution during catalytic processes, which further exert influence on the catalytic reaction. Traditional research in theoretical catalysis often rely on static computational methods, which inherently struggle to account for the dynamic nature of catalytic processes. The integration of statistical methodologies has unveiled distinctive dynamical characteristics of catalysts at different reaction states, introducing notable entropy effects that offer potential avenues for modulating reaction processes. Moving forward, our research will focus on further elucidating the intricate dynamics of catalysts and their influence on catalytic reactions. This includes establishing dynamic catalytic theories to better reveal and modify these temporal variations. Concurrently, we aim to explore the practical implications of these findings, thereby deepening our understanding of how these temporal changes affect catalytic systems. - Alloy Dynamic Catalysis
Alloy clusters are widely employed in various catalytic reactions. Different metal atoms may exhibit different interactions with reactants, which accordingly influences the dynamic behavior of catalysts during the reaction process. This may have a substantial impact on the overall catalytic efficiency. Traditional computational methods often fail to capture these dynamic changes comprehensively. By employing statistical methods, we can achieve more thorough observation of the evolution of alloy clusters during the reaction. In future research, we aim to further explore the intricate interplay between catalysts and reactants at a microscopic level, thereby providing clearer insights into reaction mechanisms and pathways. This understanding will underpin theoretical frameworks for the design and optimization of alloy clusters in catalytic applications. - Catalyst Dynamics under Confinement
The interaction between clusters and their supports is also a crucial factor influencing catalytic reactions. For example, as a common porous support, carbon nanotubes exert confinement effect on the dynamic evolution of nano-sized metal clusters, thereby modifying catalytic reaction processes. In future work, we aim to investigate more practical catalytic systems to delve deeper into the confinement effect of composite catalysts. We will also integrate these studies with dynamic catalysis theories. Ultimately, we may bridge the catalysis theory at computational-level with practical catalytic processes.
Publications
- J.-J. Sun, J. Cheng*, Solid-to-liquid phase transitions of sub-nanometer clusters enhance chemical transformation., Nat. Commun. 2019, 10, 5400. (DOI: 10.1038/s41467-019-13509-3)
- Fan, Q.-Y.; Sun, J.-J.; Wang, F.; Cheng, J.* Adsorption-Induced Liquid-to-Solid Phase Transition of Cu Clusters in Catalytic Dissociation of CO2. J. Phys. Chem. Lett. 2020, 11 (19), 7954-7959.(DOI: 10.1021/acs.jpclett.0c02499)
- Fan, Q.-Y.; Wang, Y.; Cheng, J.* Size-Sensitive Dynamic Catalysis of Subnanometer Cu Clusters in CO2 Dissociation. J. Phys. Chem. Lett. 2021, 12 (16), 3891-3897.(DOI: 10.1021/acs.jpclett.1c00506)
- Fan, Q.-Y.; Shi, Z.-H.; Wang, Y.; Cheng, J.* Charge State Dependence of Phase Transition Catalysis of Dynamic Cu clusters in CO2 Dissociation. J. Phys. Chem. C, 2021, 125 (50), 27615-27623.(DOI: 10.1021/acs.jpcc.1c08970)
- Zhang, J.#; Jiang, Y.#; Fan, Q.-Y.#; Qu, M.; He, N.; Deng, J.; Sun, Y.; Cheng, J.* ; Liao, H.-G; Sun, S.-G. Atomic Scale Tracking of Single Layer Oxide Formation: Self-Peeling and Phase Transition in Solution. Small Methods ,2021, 2001234.(DOI: 10.1002/smtd.202001234)
- Fan, Q.-Y.#; Liu, J.-L.#; Gong, F.-Q.; Wang, Y.; Cheng, J.* Structural Dynamics of Ru Cluster During Nitrogen Dissociation in Ammonia Synthesis. Phys. Chem. Chem. Phys. 2022, 24 (18): 10820-10825.(DOI: 10.1039/D2CP00678B)
- Sun, J.-J.#; Fan, Q.-Y.#; Jin, X.; Liu, J.-L.; Liu, T.-T.; Ren, B.; Cheng, J.* Size-Dependent Phase Transitions Boost Catalytic Activity of Sub-nanometer Gold Clusters. J. Chem. Phys. 2022, 156 (14): 144304.(DOI: 10.1063/5.0084165)
- Qi-Yuan Fan#, Jing-Li Liu#, Fu-Qiang Gong, Ye Wang and Jun Cheng*, Structural dynamics of Ru cluster during nitrogen dissociation in ammonia synthesis. Physical Chemistry Chemical Physics, 2022, 24, 10820-10825.(DOI: 10.1039/D2CP00678B)
- Gong, F.-Q.; Guo, Y.-X.; Fan, Q.-Y.; Cheng, J. , Dynamic catalysis of sub-nanometer metal clusters in oxygen dissociation. Next Nanotechnology, 2023, 1, 100002.(DOI: 10.1016/j.nxnano.2023.100002)
- Qi-Yuan Fan, Fu-Qiang Gong, Yun-Pei Liu, Hao-Xuan Zhu, Jun Cheng*.Modeling Dynamic Catalysis at ab Initio Accuracy: The Need for Free-Energy Calculation. ACS Catal. 2024, 14, 21, 16086–16097. (DOI: 10.1021/acscatal.4c05372)
- Qi-Yuan Fan‡, Yun-Pei Liu‡, Hao-Xuan Zhu, Fu-Qiang Gong, Ye Wang, Weinan E, Xinhe Bao, Zhong-Qun Tian and Jun Cheng. Entropy in catalyst dynamics under confinement. Chem. Sci., 2024, 15, 18303-18309(DOI: 10.1039/D4SC05399K )
- Fu-Qiang Gong, Yun-Pei Liu, Ye Wang, Weinan E, ZhongQun Tian, and Jun Cheng. Machine Learning Molecular Dynamics Shows Anomalous Entropic Effect on Catalysis via Surface Pre-melting of Nanoclusters. Angew. Chem. Int. Ed. 2024, e202405379(DOI: 10.1002/anie.202405379)

贾梦蕾 
吴倩云 
施忠浩 
李淑离 
王程玄